CBSE Class 11-science Answered
2 oxides of metal contain 50% and 40% of metal respectively. If formula of first oxide is MO then determine the formula of second oxide
Asked by anshulsingh7234 | 18 Jun, 2019, 09:56: AM
Given:
Oxide I contains 50% of M.
Oxide II ci=ontains 40% of M.
The first oxide is given as MO.
Let the atomic mass of M be 'x'.
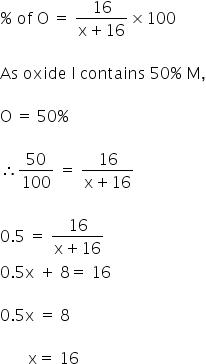
So the atomic mass of metasl is 16
Now for IInd oxide,
M = 40%
O = 60%
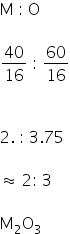
Formula of the second oxide is M2O3.
Answered by Varsha | 18 Jun, 2019, 11:25: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by jayag1298 | 08 Apr, 2024, 03:14: PM
CBSE 11-science - Chemistry
Asked by omniscientnjf2021 | 07 Apr, 2024, 10:18: PM
CBSE 11-science - Chemistry
Asked by hcnainwal | 15 Jun, 2023, 10:39: AM
CBSE 11-science - Chemistry
Asked by Jprmumal29 | 18 Dec, 2022, 09:48: PM
CBSE 11-science - Chemistry
Asked by mallikarjunasangi28 | 22 Jul, 2022, 07:57: PM
CBSE 11-science - Chemistry
Asked by vedwatisharma79 | 10 Jun, 2022, 05:27: PM
CBSE 11-science - Chemistry
Asked by thathvakunjusree | 10 Dec, 2021, 06:46: AM
CBSE 11-science - Chemistry
Asked by udheshraddha2004 | 28 Oct, 2021, 09:37: PM
CBSE 11-science - Chemistry
Asked by arunparewa2000 | 27 Oct, 2021, 06:59: PM
CBSE 11-science - Chemistry
Asked by arttameher038 | 23 Aug, 2021, 07:06: AM







