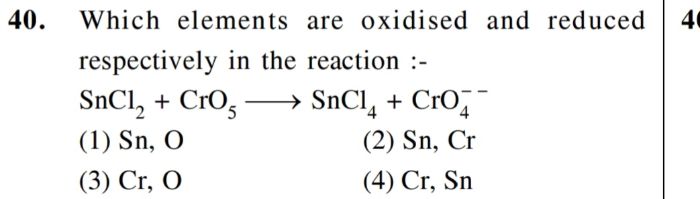CBSE Class 12-science Answered
1.how can we find the oxidation states of a compound when its electronic configuration is given?
2. what is standard electrode potential? how can we determine the reducing property of an element using its standard electrode potential?
3.how can we calculate the oxidising power of a given element?
4.what is the reason for irregular variation of ionisation enthalpy of 1st series of transition elements?
5.what is the meaning of less negative hydration enthalpy of cu+?
6.what is meaning of 2nd ionisation enthalpy?
7.how oxidation state determines stability of an element?
Asked by Karishma | 27 Sep, 2014, 02:05: PM
Dear guptakarishma54@gmail.com
Thanks for asking us a question in Ask the Expert section of TopperLearning.com.
We cannot entertain 7 different questions in a single query. In case of multiple questions within a query, please post each question individually and let us know where you are getting stuck so that we would be able to explain things better. Answer for your first query is given below:
Oxidation state is defined as the oxidation number per atom and oxidation number is the charge present on an atom of an element in combined state. So, oxidation numbers are not so much related to electron configuration.
But sometimes electron configuration of an atom tell us the number of electrons in the outer energy level i.e., the valence shell electrons. The oxidation number is derived from the number of valence shell electrons.
Eg: If the valence shell has 3 valence electrons, then we could figure out that the oxidation numbers have to be 3+ or 5- because we know that if we take away 3 electrons or add 5 electrons, the atom would reach an octet.
Regards,
Topperlearning Team.
Topperlearning Team.
Answered by Arvind Diwale | 29 Sep, 2014, 01:42: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by anubhutiupadhaya | 27 Feb, 2024, 04:28: PM
CBSE 12-science - Chemistry
Asked by basib61203 | 08 Feb, 2024, 06:03: PM
CBSE 12-science - Chemistry
Asked by ABHILASHA | 04 Mar, 2021, 02:26: AM
CBSE 12-science - Chemistry
Asked by ghoshmahadev037 | 20 Sep, 2020, 11:45: AM
CBSE 12-science - Chemistry
Asked by mufeedatvp2000 | 20 Apr, 2020, 02:53: PM
CBSE 12-science - Chemistry
Asked by patra04011965 | 25 Sep, 2019, 10:22: PM
CBSE 12-science - Chemistry
Asked by patra04011965 | 22 Sep, 2019, 01:42: PM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 30 Aug, 2019, 08:09: AM
CBSE 12-science - Chemistry
Asked by Debdulal | 29 Aug, 2019, 04:46: PM
CBSE 12-science - Chemistry
Asked by Debdulal | 29 Aug, 2019, 04:44: PM